 Wojtek is the main author of a new paper published in J. Phys. Chem. C on hydrogen peroxide generation at liquid|liquid interfaces. This is the latest in a series of papers (e.g. 1-5) about various reactions at the interface between immiscible liquids published in collaboration with Hubert Girault’s group in Lausanne.
Wojtek is the main author of a new paper published in J. Phys. Chem. C on hydrogen peroxide generation at liquid|liquid interfaces. This is the latest in a series of papers (e.g. 1-5) about various reactions at the interface between immiscible liquids published in collaboration with Hubert Girault’s group in Lausanne.
Reactions at liquid|liquid interfaces are interesting from a fundamental science perspective, as such interfaces exist in many technological and biological systems, but also because such reactions can be useful in industrial processes. One example could be that reactions can take place at the interface, with the reactants in one phase and the product going into the other phase, potentially saving on purification of the end product.
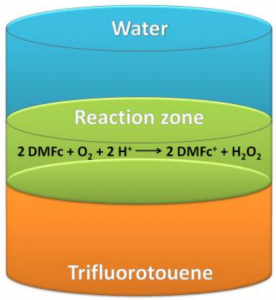
In this manuscript the reaction is hydrogen peroxide generation. It’s a deceptively simple reaction, but as usually the difficulties are in the details. With some 2 million tons of H2O2 produced every year, generation of hydrogen peroxide is an important industrial process and ways of lowering the cost of production could be very valuable. Girault has been investigating H2O2 generation at liquid|liquid interfaces for a long time now, experimenting with different solvents, catalysts, redox probes, etc. In this paper we have looked at what is likely the simplest possible* biphasic system for H2O2 generation, since it contains only the solvents, the reactants (in this case dissolved oxygen and protons from an acidic solution), and an electron donor (DMFc). DMFc gives off an electron to compensate for the positive charge of the protons to create the neutral H2O2 molecule. Without this donor the system doesn’t work. So, removing any of the components would stop the H2O2 generation, which is why we consider this as the simplest system for this process. However, it’s slow and not very efficient, so from an industry perspective finding a good, cheap and reusable catalyst is still necessary.
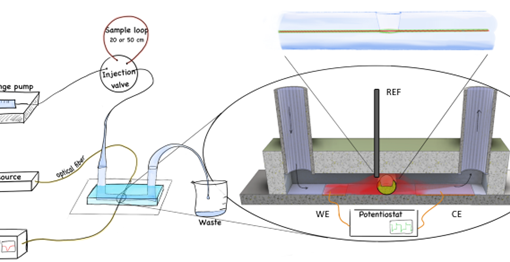
Here we used a scanning electrochemical microscope tip to detect the generated hydrogen peroxide and measure how much is created. The results from the SECM measurements were compared with some simple computer models to see if we could say something about the reaction process. We also measured the partitioning of H2O2, and found that about 3% of the peroxide escapes into the organic solution.
* The title of the manuscript was originally “The simplest liquid-liquid…”, but the editor didn’t like that.
1. Jedraszko, J., Nogala, W., Adamiak, W., Girault, H. H. & Opallo, M. Scanning electrochemical microscopy determination of hydrogen flux at liquid|liquid interface with potentiometric probe. Electrochem. commun. 43, 22–24 (2014).
2. Adamiak, W. et al. Hydrogen and Hydrogen Peroxide Formation in Trifluorotoluene–Water Biphasic Systems. J. Phys. Chem. C 118, 23154–23161 (2014).
3. Jedraszko, J. et al. Hydrogen Peroxide Generation at Liquid|Liquid Interface under Conditions Unfavorable for Proton Transfer from Aqueous to Organic Phase. J. Phys. Chem. C 117, 20681–20688 (2013).
4. Jedraszko, J. et al. Catalysis at the room temperature ionic liquid|water interface: H2O2 generation. Chem. Commun. 51, 6851–6853 (2015).
5. Hidalgo-Acosta, J. C. et al. Catalysis of water oxidation in acetonitrile by iridium oxide nanoparticles. Chem. Sci. 6, 1761–1769 (2015).

One thought on “A Simple Liquid–Liquid Biphasic System for Hydrogen Peroxide Generation”