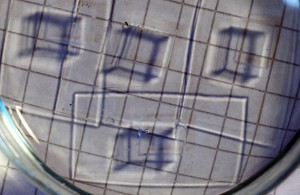
We just had a new paper published in Microfluidics and Nanofluidics concerning the compatibility of some organic solvents with PDMS-based microfluidic devices. In a well-known study (cited ~1600 times) the Whitesides-group argued that how much PDMS swells in a solvent is a good indication of how compatible that solvent is with PDMS-based devices. They also show that swelling is closely related to the Hildebrandt solubility parameter, δ, which is a measure of the cohesive energy density of the solvent. If δ for the solvent is close to that of PDMS (δPDMS = 7.3 cal1/2 cm−3/2) the swelling of PDMS is large and the solvent is deemed incompatible with PDMS-based devices.
I our study we performed electrochemical measurements over a period of 4 hours in a PDMS-based microfluidic device in several organic solvents frequently used in electrochemistry. We measured the swelling of PDMS in a few solvents not included in the Whitesides study and confirmed that δ and swelling are closely related. However, we saw no correlation between swelling and incompatibility with electrochemical measurements. We could record stable voltammograms over a four hour period even using trifluorotoluene as a solvent, where the selling was more than 40%.
Thus, our conclusion was that δ is not a good measure for PDMS compatibility.
The paper is available as open access here from Springer, or directly as pdf here.