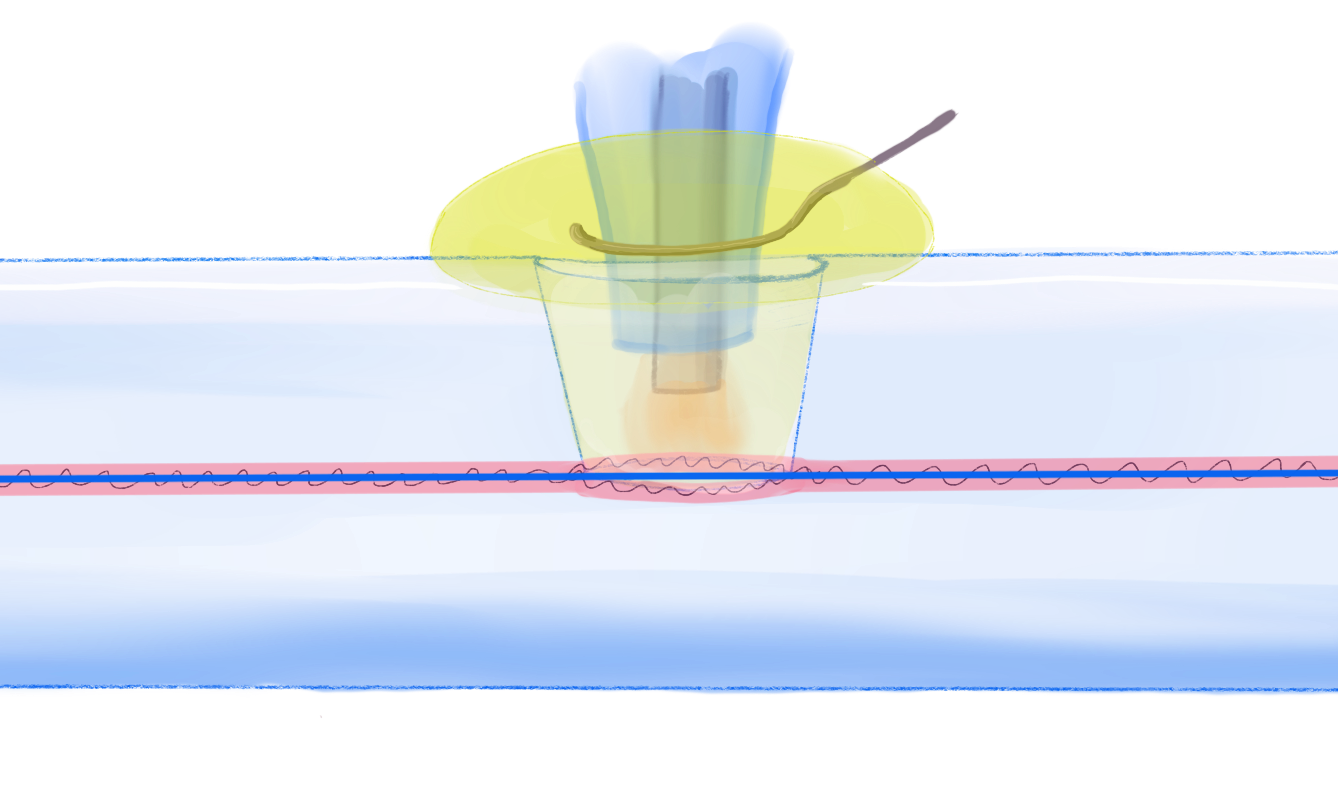
We just published an article1 combining electrochemistry and interferometry in a microcavity. This work was done in collaboration with colleagues from Warsaw University of Technology and the National Institute of Telecommunications, who are responsible for the optics part of the work.
The experiment is based on a microcavity in-line Mach-Zehnder interferometer (µIMZI), which is a small cavity drilled into an optical fibre using femtosecond laser-ablation. The cavity goes partly into the core of the fibre and light travelling through the fibre will be split between continuing in the core and passing through the cavity. If we change the refractive index in the cavity the optical path changes and this difference can be measured as the light from the two paths interfere. The sensitivity is very high – changes in refractive index in the fifth decimal can be measured.
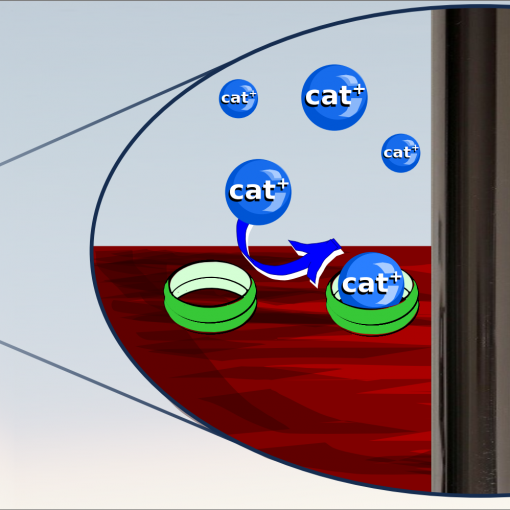
Here, we put a microelectrode into the cavity and used it to oxidise and reduce a redox probe (in this case ferrocenedimethanol) and followed how the electrochemical reaction leads to changes in the optical properties of the liquid in the cavity. The changes are minute – only about 0.00007 RIU – but still visible in the cavity interferometer even if no difference was seen NIR-spectroscopy.
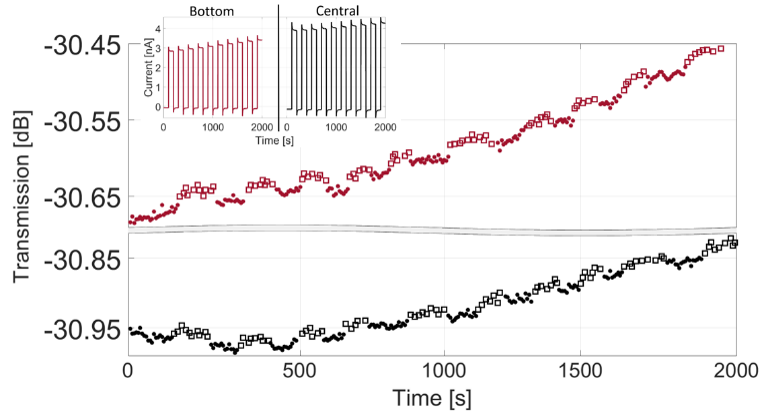
The measurements were supported by calculations both of the electrochemical and of the optical response. We from this we see that the light is split into two paths, with the one through the cavity located about 5 µm above the floor of the cavity. Therefore, we get a larger response when the electrode is deep inside the cavity, even if the electrochemical current is lower.
These are the first measurements of its kind, combining a µIMZI with electrochemistry. We actually didn’t know if it was possible at all to do these measurements when we started, so this was a nice proof-of-concept. Now to think about what to use it for.

One thought on “Electrochemistry in an optical fiber microcavity – optical monitoring of electrochemical processes in picoliter volumes”