Alzheimer’s Disease (AD) is one of the most common neurodegenerative diseases. The first time it was described 100 years ago; however, up to now, it is mysterious. According to the most accepted theory, development of this disease is connected to the interactions of copper ions with peptides called beta-amyloids. The best known beta-amyloid is Aβ1–16 but according to new findings in Aβ deposits in brains of sporadic and familial AD victims also detected Aβ4–42 and Aβ5–42.
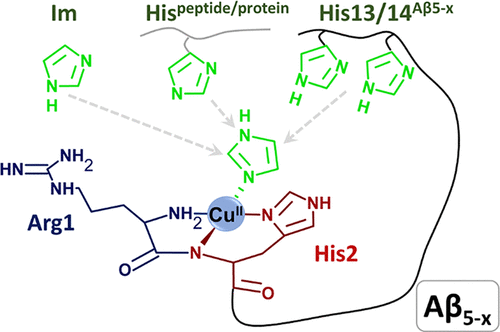
The electrochemical aspects of Cu(II)-Aβ interactions were part of Magdalena Wiloch’s PhD topic. Now, some years after her defence, a new article titled “Aβ5–x Peptides: N-Terminal Truncation Yields Tunable Cu(II) Complexes”1 is published. As interactions of newly discovered amyloids with copper ions are quite a complicated issue therefore not only electrochemical techniques (voltammetry, potentiometry) were used but also spectroscopic methods like UV−vis, CD, and fluorescence spectroscopy.
Magda’s part of this work reveals that Aβ5–x is a special peptide because cooper ions can undergo reductions from Cu(II) to Cu(I) but also oxidation from Cu(II) to Cu(III). It is unusual because when Aβ1–16 binds Cu(II) only the reduction process appears and when Aβ4–16 binds these cations only the oxidation process can take place.
The research work carried out by Magdalena is now continued by the research group in which she was doing her PhD.
- N. E. Wezynfeld, A. Tobolska, M. Mital, U. E. Wawrzyniak, M. Z. Wiloch, D. Płonka, K. Bossak-Ahmad, W. Wróblewski, W. Bal
Aβ5–x Peptides: N-Terminal Truncation Yields Tunable Cu(II) Complexes, Inorg. Chem. (accepted). (link – open access)




